An electron octet is not always stable!
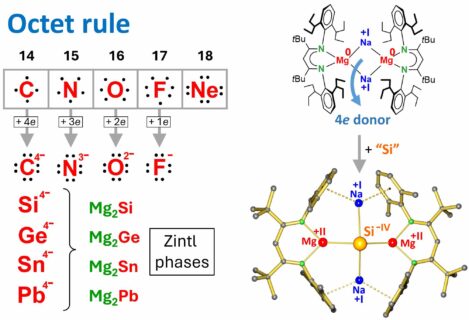
It is taught at an early stage that main group atoms strive to collect eight electrons in their valence shell. This octet rule is the origin for the total inertness of the element Ne. It also explains the high stability of F– and O2– anions. However, when charges get higher the anions rapidly become much more reactive. Highly charged N3– and especially C4– are rare and highly reactive. The same is true for the heavier group 14 anions Si4–, Ge4–, Sn4– and Pb4–. Magnesium silicide (Mg2Si) can be obtained by burning magnesium metal in sand (SiO2) at temperatures over 3000 °C. The charge on the Si4– anion in Mg2Si is so high that the electrons are partially delocalized over Mg. It is a narrow-gap semiconductor with photovoltaic and thermoelectric applications.
The Harder group at FAU has now reduced Si to Si4– at room temperature [1]. The key to this very facile reaction is an earlier reported soluble form of magnesium metal [2]. The Si compound can be seen as a molecular Zintl phase. Although Si4– follows the octet rule, it is extremely reactive and immediately inserts in a very strong C-H bond. However, the same complexes with heavier Sn4– and Pb4– anions are more stable and could be detected. The work has been published in the renowned journal Nature Synthesis [1].
Further information
[1] Nat. Synth (2026). https://doi.org/10.1038/s44160-026-01006-7
[2] Nature 592, 717–721 (2021). https://doi.org/10.1038/s41586-021-03401-w
Contact
Prof. Dr. Sjoerd Harder
Chair of Inorganic and Organometallic Chemistry