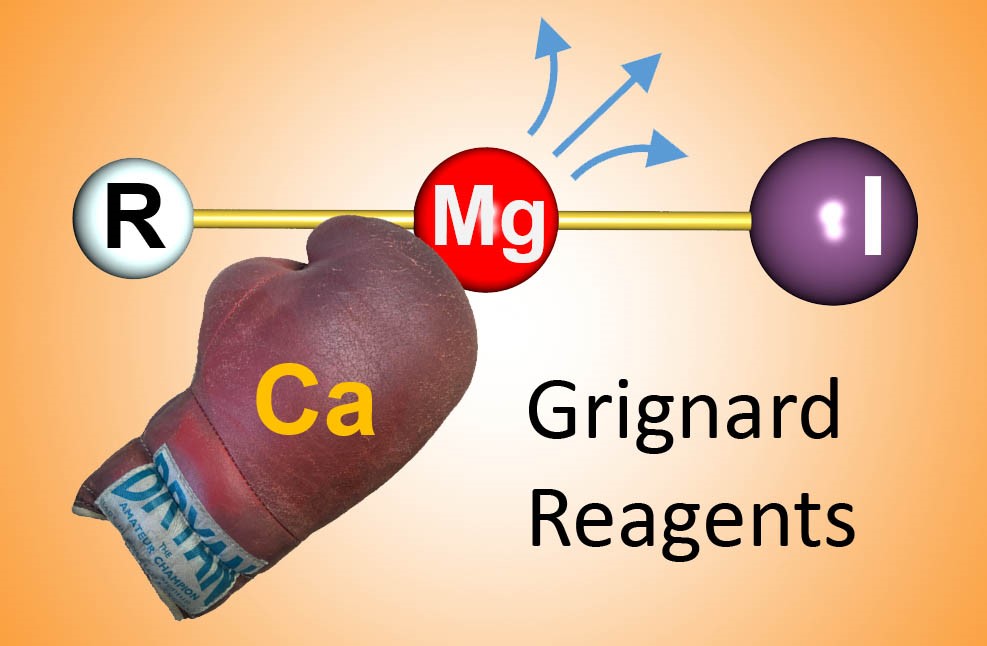
Grignard reagents are by far the most well-known organometallic compounds. Although more than hundred years old, these reagents are still today essential in organic synthesis and large scale industrial processes. These complexes are based on the metal magnesium but there is increasing interest in replacing magnesium for its heavier neighboring metal calcium. Magnesium may be an abundant metal but since 85% of the world’s supply is produced in China, it is also a critical metal with supply chain risks.
In addition, calcium Grignard reagents are far more reactive and show unusual reactivity which today has never been fully exploited. As the calcium metal itself is hardly reactive, it has been found a tightrope walk to produce calcium Grignard reagents. Dr. Jens Langer, who has been involved in the very first structural characterizations of calcium Grignard’s, and Prof. Sjoerd Harder now report on the latest developments in this area in Nature Chemistry Reviews [1].
This includes the recent discovery that calcium Grignard reagents can simply be made by grinding the metal with an organic compound. Grinding with mortar and pestle is one of the oldest methods to achieve chemical reactions by friction. It is amusing that stone age methods are the key to modern chemistry. These calcium reagents are not just more reactive but also found application in catalysis [2] and show rather unusual reactivity which normally can only be achieved with noble transition metals [3]. A unique characteristic of the transition metals is the availability of d-orbitals.
Harder and Langer therefore discuss the strange highly controversial possibility that, contrary to general believe, calcium may also use d-orbitals in its chemistry. This comprehensive review article is accessible using the link below or can be requested by contacting the authors.
Further information:
Link: https://rdcu.be/dqvn4
- [1] S. Harder, J. Langer, Nature Chemistry Reviews https://doi.org/10.1038/s41570-023-00548-0
- [2] Early Main Group Metal Catalysis, Editor: Sjoerd Harder, Wiley-VCH, Weinheim, Germany, 2020.
- [3] B. Rösch, T. X. Gentner, J. Langer, C. Färber, J. Eyselein, L. Zhao, C. Ding, G. Frenking, S. Harder, Science 2021, 371, 1125.
Contact:

Prof. Dr. Sjoerd Harder
Chair of Inorganic and Organometallic Chemistry
Professors