Tailored Thermal Switches: Zinc Ferrite Nanoparticles with Tunable Curie Temperatures
Heat at the push of a button – but only up to a precisely defined temperature. What may initially sound like a technical detail is crucial for many applications: in cancer therapy, healthy tissue must not overheat, while in polymer-based materials, adhesive bonds should be selectively released without damaging surrounding structures. Researchers at the Chair of Particle-Based Materials Chemistry led by Prof. Karl Mandel at the Department of Chemistry and Pharmacy at FAU now demonstrate how magnetic nanoparticles can be designed as self-regulating thermal switches – with freely adjustable temperature limits ranging from room temperature up to 250 °C.
An Intrinsic Safety Mechanism
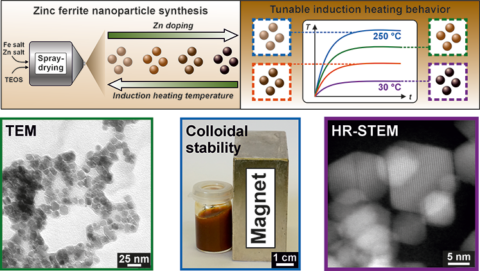
At the center of the study are iron oxide nanoparticles with zinc in their structure, so-called zinc ferrites (ZnₓFe₃₋ₓO₄), whose key property lies in their Curie temperature. This temperature marks the transition from a magnetically ordered to a paramagnetic state. Once reached, the particles lose their magnetic order – and with it their ability to generate further heat in an alternating magnetic field. The temperature automatically reaches a plateau; exceeding it is physically impossible.
“We deliberately use the Curie temperature as an intrinsic material safety mechanism,” explains Leoni Luthardt. “The system regulates itself – regardless of how strongly we excite it externally.”
Since the temperature achieved during inductive heating typically depends on many external parameters – such as field strength, heat dissipation, geometry, or environment – such an intrinsic limitation provides a significant advantage.
Two Parameters for Precise Temperature Design
The researchers combine a scalable spray-drying synthesis with a subsequent targeted high-temperature treatment between 1000 and 1100 °C. Two parameters are decisive: the zinc content within the particle structure and the post-synthesis annealing temperature.
As the zinc content increases, the Curie temperature decreases. At the same time, higher annealing temperatures lead to higher achievable heating temperatures. In this way, maximum induction heating temperatures can be tuned continuously between approximately 30 °C and 250 °C. Notably, unusually high zinc substitution levels of up to x = 0.75 could be stably incorporated into the structure. The magnetic properties can therefore be directly controlled via the chemical composition.
A System Covering Broad Temperature Windows
The wide range of adjustable temperatures makes the system particularly versatile. Particles with low zinc share reach high heating temperatures and are therefore suitable for technical applications such as inductive curing or debonding-on-demand. Highly zinc-containing variants, in contrast, operate specifically in the mild temperature range relevant, for example, to magnetic hyperthermia.
“We can define the maximum heating temperature already at the material level. This opens up entirely new degrees of freedom for applications in which temperature limits must be strictly maintained,” says Karl Mandel.
Stable, Scalable, and Cobalt-Free
The nanoparticles also exhibit high colloidal stability in aqueous dispersion and can be reliably heated inductively. They combine good dispersibility with controlled heat generation – a combination that is far from trivial in practice.
Importantly, the system operates entirely without cobalt. Instead, zinc is used, which is more abundant, cost-effective, and biocompatible.
By combining scalable synthesis, a broad temperature window, and intrinsic overheating protection, the study demonstrates how magnetic nanoparticles can be rationally designed as tailored heating materials – for both technical processes and medical applications. The article was recently published in the journal Small and is available at the following link: https://onlinelibrary.wiley.com/doi/10.1002/smll.202513314?af=R