Nanographene: Aggregation and Size-Dependent Photophysical Behavior
When the graphene lattice is divided into smaller, well-defined segments, so-called nanographenes and graphene nanoribbons are obtained. These structures bridge the gap between the molecular level of carbon-rich systems and the realm of carbon-based materials. The targeted synthesis of nanographenes and nanoribbons with precise size and carbon atom count is, in principle, only achievable through a bottom-up approach, in which increasingly larger architectures are constructed from smaller molecular building blocks. This strategy enables the preparation of systematic series of nanographenes, allowing for a direct correlation between molecular structure and physical properties—an essential prerequisite for potential applications. However, the limited solubility of larger nanographenes and nanoribbons often leads to aggregation and the formation of poorly soluble species, which severely hampers detailed characterization and the establishment of reliable structure–property relationships.
The research groups of Professor Dirk M. Guldi and Professor Norbert Jux (both FAU Erlangen), together with Professor Fabrizia Negri (Bologna), have now successfully addressed this challenge. They achieved the synthesis of a fully benzenoid nanographene, consisting exclusively of six-membered rings (NG-Br), as well as its porphyrin conjugate (NG-Por), through a cascade of Suzuki cross-coupling reactions, Sandmeyer-type brominations, and Scholl oxidations. Of particular interest was the determination of the concentration at which nanographenes begin to aggregate, as the photophysical properties of aggregates typically differ markedly from those of isolated monomers. For NG-Br, an aggregation constant exceeding 10⁶ M⁻¹ was determined. Computational analyses suggest that aggregation initially leads to the formation of a dimer with parallel-oriented carbon frameworks. Consequently, ultralow concentrations on the order of 10⁻⁸ M⁻¹ were employed to investigate the intrinsic photophysics of the monomeric species.
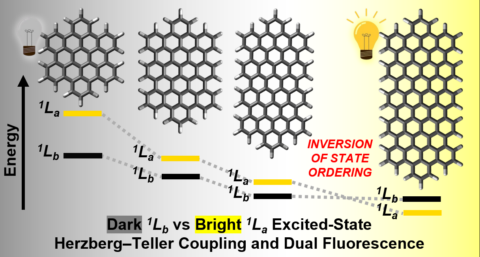
Comprehensive spectroscopic studies, supported by advanced theoretical calculations, revealed a subtle energetic convergence of the (S1) / (Lb / α-band) and (S2) / (La / p-band) excited states, giving rise to pronounced vibronic coupling mediated by the Herzberg–Teller mechanism. This effect manifests clearly in the fluorescence spectra and is corroborated by time-dependent density functional theory (TDDFT) calculations. These findings also resolve previously reported discrepancies in the absorption and fluorescence spectra of related nanographenes. Furthermore, a strong vibronic mixing of the (S1,1) and (S2,0) states was observed, leading to anti-Kasha emission behavior. In NG-Por, an ultrafast (S1–S1) energy transfer from the nanographene moiety to the porphyrin unit was detected.
Overall, these results demonstrate the presence of strong vibronic effects in extended carbon architectures and provide compelling evidence of how the length and size of nanographenes and graphene nanoribbons critically influence their photophysical properties, thereby uncovering fundamental structure–property relationships. This knowledge enables the rational design of tailored molecular systems for applications in areas such as single-electron transistors, organic light-emitting diodes (OLEDs), and organic solar cells.
Original Publication:
Giovanni M. Beneventi, Filip Božinović, Phillip Greißel, Max M. Martin, Frank Hampel, Alejandro Cadranel, Fabrizia Negri, Norbert Jux, and Dirk M. Guldi
“Synthesis and Excited-State Dynamics in Molecular Nanographene: Herzberg–Teller Vibronic Coupling and Energy Transfer to Porphyrins”
https://doi.org/10.1021/jacs.5c14685