Cumulenes: Beauty in a Line
The chemical world of elemental carbon modifications—carbon allotropes—has grown remarkably diverse, extending far beyond the naturally occurring forms of diamond and graphite. In addition to these three-dimensional structures, synthetic nanostructures such as quasi-one-dimensional carbon nanotubes, spherical fullerenes, and two-dimensional graphene, along with their chemical derivatives, have now been realized in the laboratory. Each of these allotropes exhibits unique and highly interesting physical and chemical properties, which has made carbon modifications and carbon-rich compounds a major focus of global research over the past decades.
However, a close inspection of the carbon family reveals a conspicuously missing member: the truly one-dimensional carbon chain. This structure would consist of carbon atoms in a single linear arrangement, representing an ultimate molecular wire of potentially “infinite” length, also known as carbyne. From a chemical perspective, two distinct structural motifs are theoretically possible: one in which single and triple bonds alternate (polyynes) and another consisting of consecutive double bonds (polycumulenes). These two configurations correspond to the quantum-mechanically predicted forms of the one-dimensional carbon chain. The former exhibits semiconducting properties, with a band gap determined by the alternating C–C bond lengths, whereas the latter displays metallic behavior due to the uniform bond lengths of the consecutive double bonds. Both configurations are additionally predicted to possess exceptional tensile strength and outstanding electrical conductivity.
In practice, however, free carbyne has not yet been isolated in the laboratory. To explore its properties, researchers have turned to the synthesis of shorter carbon nanowires as molecular analogs: oligoynes (polyynes) and cumulenes (polycumulenes). These model systems already display fascinating physicochemical characteristics. While oligoynes with chain lengths exceeding 30 alkyne units have been successfully synthesized, characterized, and studied with respect to their physical and optical properties, cumulenes have remained comparatively underexplored: until now, cumulene chains with a maximum of only nine consecutive C=C bonds have been isolated, and knowledge of their optical and photophysical properties has been extremely limited.
A significant advance has now been made by the research group of Professor Dirk M. Guldi, in collaboration with the teams of Professor Rik R. Tykwinski, Professor Fabrizia Negri, and Professor Matteo Tommasini. In this interdisciplinary effort spanning synthetic, physical, and theoretical chemistry, odd-numbered cumulenes containing up to seven consecutive double bonds were spectroscopically analyzed for the first time, and their excited-state deactivation pathways were elucidated. These studies revealed a complex interplay between nuclear degrees of freedom and electronic properties, demonstrating that the photoinduced dynamics can be precisely controlled through conformational effects such as rotational freedom along the chain.
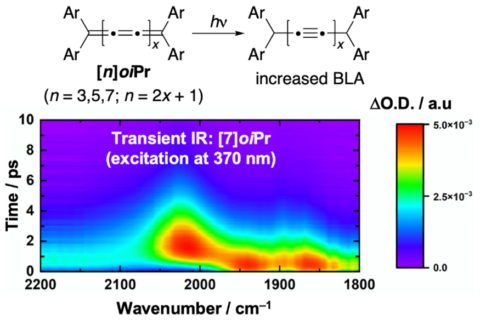
Moreover, electronic excitation through light absorption induces a transient alteration of the C–C bond characteristics within the carbon nanowire. The chain undergoes a brief photoisomerization from the original cumulene arrangement with uniform bond lengths toward an oligoyne-like configuration with alternating bond distances. This short-lived transformation opens highly promising avenues for photonics applications, as it enables the controlled modulation of electronic communication along the cumulene nanowire using light.
This work represents a key step toward understanding the optical properties of cumulenes and the carbon allotrope carbyne/polycumulenes, laying the foundation for future developments in molecular wires and light-controlled nanotechnologies.
Original Publication:
T. Ullrich, B. Sun, Y. Yang, C. M. Schüßlbauer, M. J. Ferguson, D. Fazzi, F. Negri, M. Tommasini, R. R. Tykwinski, D. M. Guldi, J. Am. Chem. Soc. 2025, 147, 21419–21431
Excited State Dynamics of Odd [n]Cumulenes: Chain Length and Conformational Effects
doi.org/10.1021/jacs.5c00147