Research
Challenge:
New materials are the key to emerging technologies. Complex nanomaterials are essential for better catalytic processes, and improved energy conversion and energy storage technologies for renewable energies. Also, in the field of molecular electronics and sensor technology new nanomaterials are at the heart of innovation.
Most of the fascinating functionalities that we encounter in these complex materials arise from their interface properties. The conversion of light, chemical energy and electricity requires the transport either of electrons, of ions, of atoms, or of molecular species via phase boundaries. Building complex nanostructured materials, we can improve the efficiency of these processes or even implement entirely new functionalities. Up to date, however, the synthesis of such complex nanomaterials mostly relies on empirical trial-and-error approaches. With the materials getting more and more complex, progress is slowing down as can already be seen in many areas of nanotechnology. The key to eliminate the ‘materials bottleneck’ is the knowledge-based development of new materials: We need to understand how transport and transformation processes work at the microscopic level, which factors limit their performance and, very importantly, why certain nanomaterials undergo degradation. Finally, we need to transfer this knowledge to the development of new devices.
Strategy and Mission:
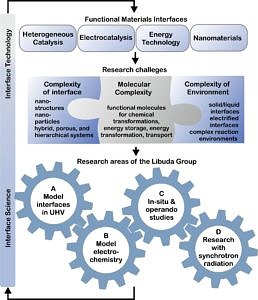
Starting from this challenge, the research mission of our group aims at
- understanding chemical transformations and physical properties of complex interface-controlled materials at the molecular scale and at
- transferring this knowledge to real materials and real application conditions.
To this end we follow a three-step research strategy:
(I) Model approach to interface-controlled materials
Real materials and devices are outstandingly complex systems, both from a structural and from a chemical point of view, thereby preventing us from obtaining atomic-level insights in many cases. Model systems are the key to understand nanomaterials and their interfaces at the microscopic level. The idea of the model approach involves chemical or structural complexity introduced in a controlled fashion and under ultraclean conditions. This allows us to derive structure-functionality relationships at interfaces and nanostructures with atomic-precision control. In a second step, this knowledge can be transferred to real materials and conditions.
(II) Molecular and material complexity to build functional interfaces
Functionalities of novel interface-controlled materials rely on interfaces between complex solid materials, complex molecular materials, liquids and reactive gases. Our research aims at building and characterizing such interfaces and at developing the spectroscopic tools to study them from ideal to real conditions.
(III) Connecting ideal and real systems or conditions
A major challenge is the transfer of knowledge from ideal model experiments to real materials and application conditions. To this end, we develop and adapt spectroscopic and microscopic methods to perform in-situ spectroscopy under ambient, high pressure and electrochemical conditions. We are performing electrochemical, spectroelectrochemical and photospectroelectrochemical experiments on both ideal single crystals and real electrocatalysts and electrode materials, as well as operando spectroscopies on ideal and real nanomaterials from ultrahigh vacuum to high pressure conditions.
